The intent is for the program to provide Associates with deep, foundational experience in key Quality and Regulatory areas and create well-rounded professionals.
The Q&R FLEX Program is a two-year, multi-site rotational program for new graduates in foundational Q&R Areas.
The intent is for the program is to provide Associates with deep, foundational experience in key Quality and Regulatory areas and create well-rounded professionals.

Discover your own path
Participants will build strong business acumen and technical skills, while gaining analytical perspective in the core functions critical to the commercialization of Genentech products.

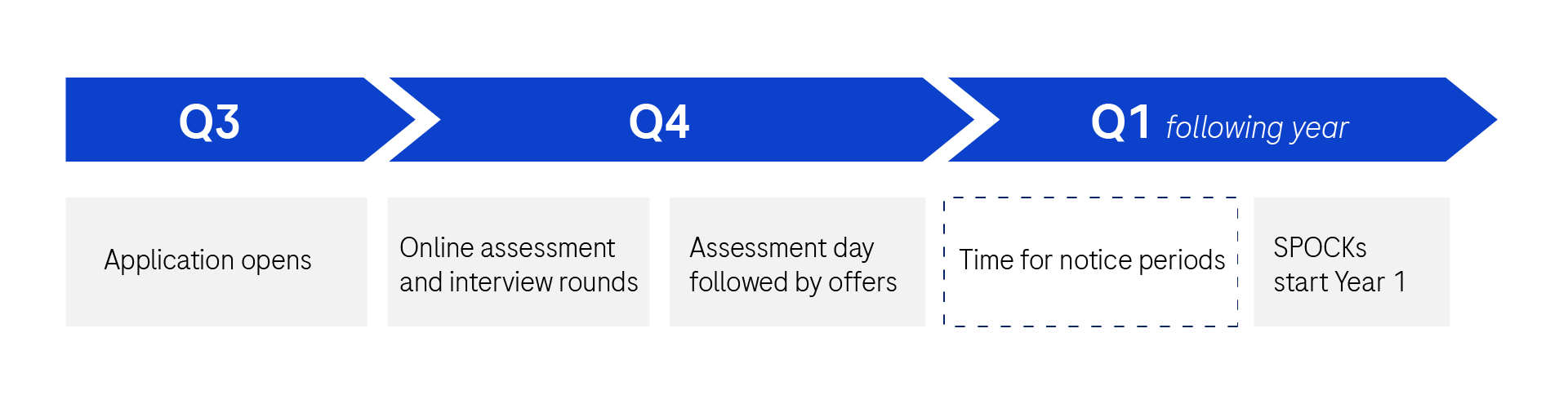
Recruitment Timelines
Applications open August/September. Offers extended By end of Q4 (October at the latest). Start in May