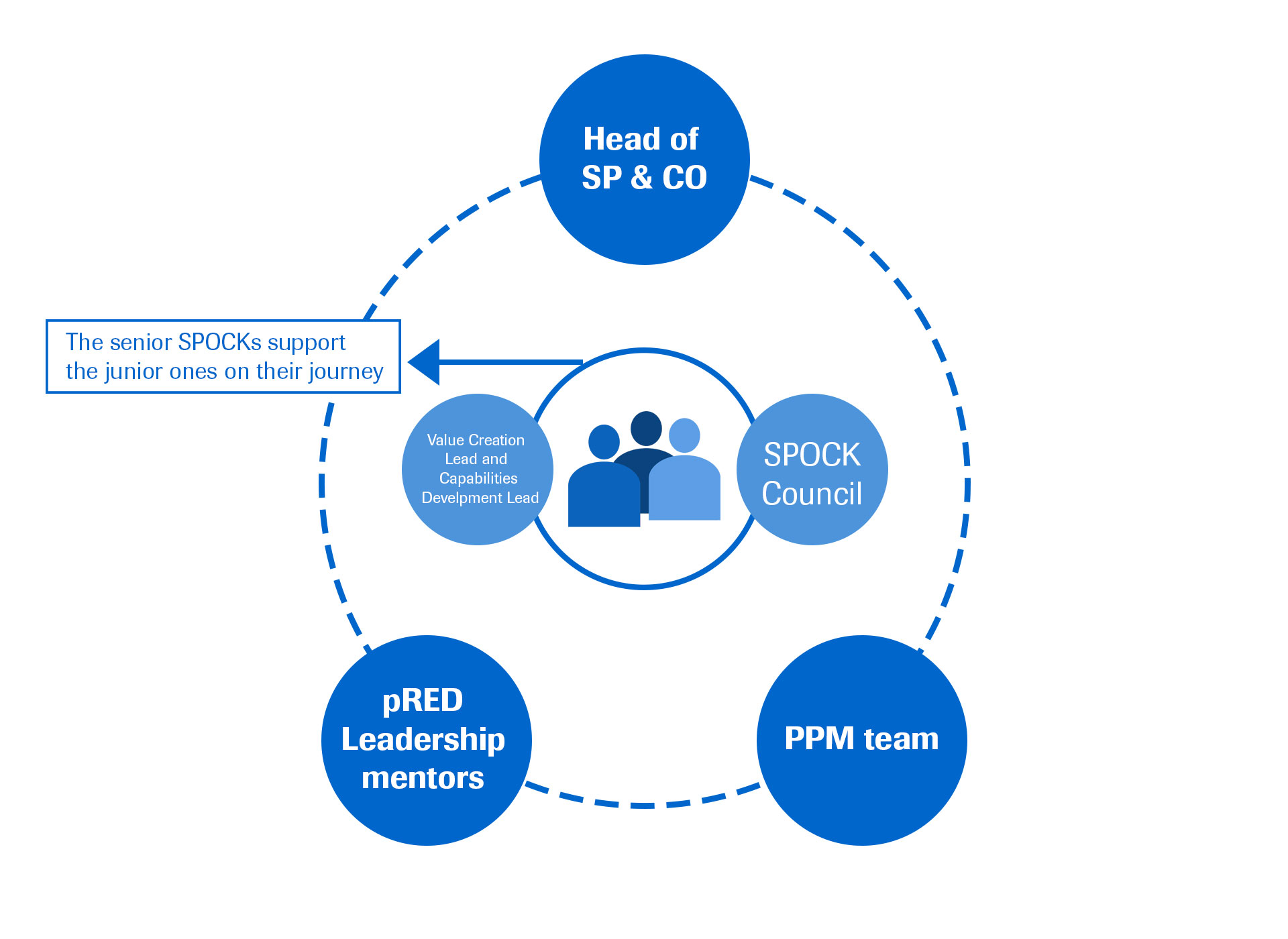
Roche Global Quality

The Global Q&R Internship Program is a two summer rotational program which provides students with the unique opportunity to gain valuable experience within Quality & Regulatory Affairs functions across Roche sites.
All intern projects are designed to have high business impact as well as serve to assess the intern’s performance and skills required for potential full-time hire following graduation.
Quality and Regulatory functions such as Design Quality Assurance, Global Safety and Recall Management, Regulatory Policy and Intelligence, Case Investigation and Resolution
What's in it for you
All intern projects are designed to have high business impact as well as serve to assess the intern’s performance and skills required for potential full-time hire following graduation.

Examples of work include:
Short-term project-based learning opportunities including pilots and first iterations, for example: process improvements, assistance with low risk submissions and technical documentation, collaborative projects with global and cross-functional teams, creation of dashboards and tools to optimize processes and procedures.
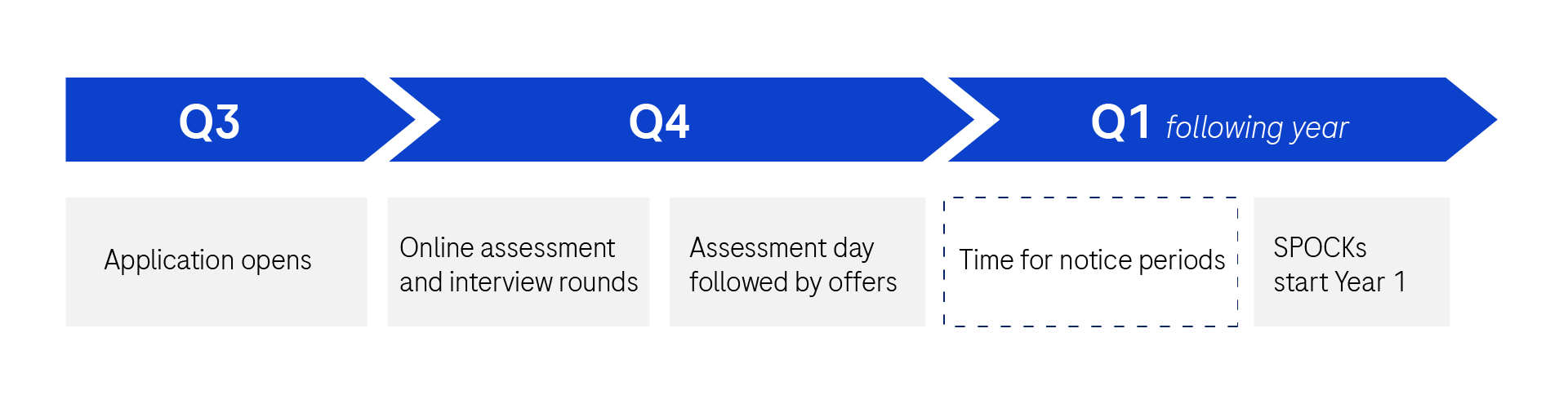
Recruitment Timelines
Applications open in September/October. Offers completed by December/January
FAQ
FAQ
